 
Some molecules at the surface, however, will have sufficient kinetic energy to escape from the liquid and form a vapor, thus increasing the pressure inside the container. The standard molar entropy, S m ° (g, 298.15 K), of gaseous glycerol (see Table 6) was calculated by adding the entropy in the liquid state, S m ° (l, 298.15 K) 12, and the molar entropy of vaporization l g S m ° derived in this work. Chemical Compounds Entropy of Vaporization 100 Azeotropic Mixture 72 Boiling Point 62 Acetic Acid 43 Alcohol 34 Pure Substance 27 Ternary Mixture. First, why the vaporization entropy is almost constant for clusters. Thermodynamic properties of R22, like vapor volume, enthalpy and entropy at. We think it is challenging to attempt to understand why this happens. During evaporation the temperature and pressure are linked to each other. This rule 1 is very useful to get a quick estimate of the heat of vaporization of a compound. Δ G v a p = Δ H v a p − T v a p × Δ S v a p = 0 Īccording to Trouton's rule, the entropy of vaporization (at standard pressure) of most liquids is about 85 to 88 J mol -1 K -1.\)), the initial pressure above the liquid is approximately zero because there are as yet no molecules in the vapor phase. In this paper we try to survey in depth a well-known rule in the discipline of Thermodynamics Trouton’s rule. 
In a phase transition such as vaporization, both phases coexist in equilibrium, so the difference in Gibbs free energy is equal to zero. Vaporization (or Evaporation) the transition of molecules from a liquid to a gaseous state the molecules on a surface are usually the first to undergo a phase change. Calculate the molar entropy of vaporization (delta Svap) 598 J/K x mol 68.6 J/K x mol 75.2 J/K. Solve the equation (specific heat at constant pressure Cp 4.1818 kJ/Kkg). Define final and initial temperature: Tf 20 ☌, Ti 100 ☌. 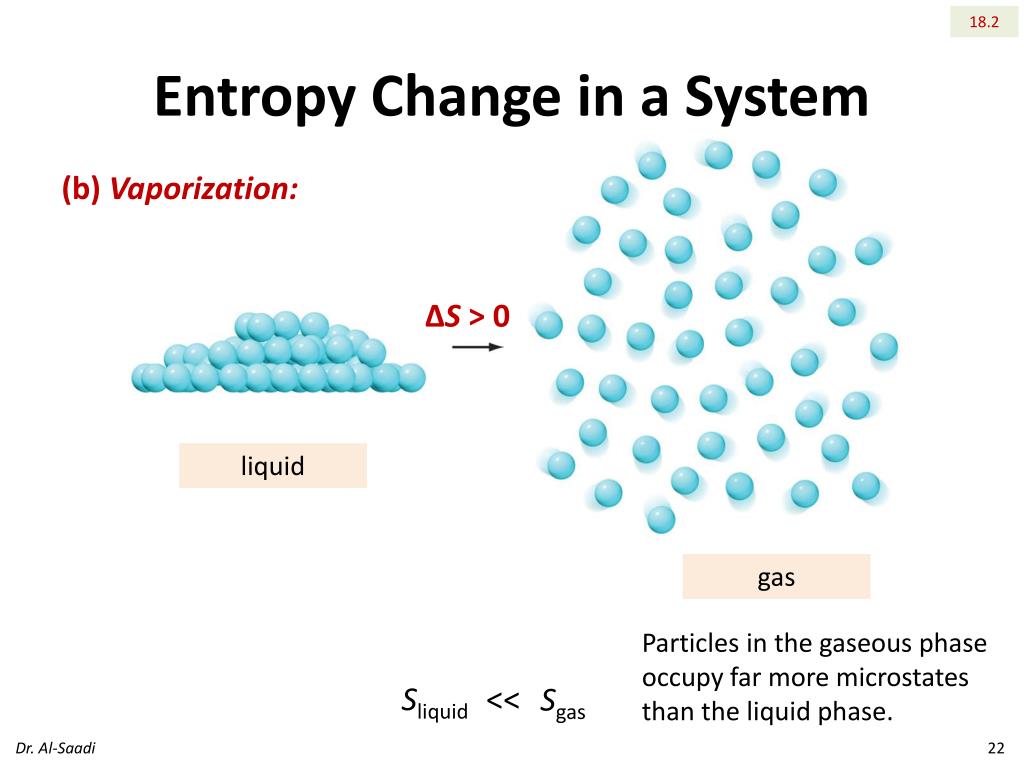
At standard pressure P o = 1 bar, the value is denoted as Δ S o vap and normally expressed in J mol -1 K -1. The entropy change on vaporization (delta Svap) of a compound or element is -Always negative-Always positive-Sometimes is positive and sometimes is negative 89.0 J/K x mol HI has a normal boiling point of -35.4 C, and its delta Hvap is 21.16 kJ/mol. We will use the change in entropy formula: s Cp × ln (Tf / Ti), where Tf and Ti indicate the final and the initial temperature, respectively. This is always positive since the degree of disorder increases in the transition from a liquid in a relatively small volume to a vapor or gas occupying a much larger space. It states that the entropy of vaporization at one atmospheric pressure of many liquids is almost constant: Table 1 shows a few values of the entropy of. What is the boiling point(C) of a liquid with entropy of vaporization 68.3 JK1mol1 and enthalpy of vaporization 35.65 kJ mol1: rounded up to two decimal. The entropy of vaporization is the increase in entropy upon vaporization of a liquid. Entropies Equation of state Internal energies Isobars Isochores Isotherms Joule - Thomson inversion Latent heats of vaporization Melting. 
Using an entropy balance, theTo calculate a global average temperature. Template:Distinguish Template:Unreferenced stub The Equation Q mHv How much energy does it take to vaporize 10 g of water. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
